REVIEW ARTICLE | https://doi.org/10.5005/jp-journals-10084-12156 |
Lipodystrophy Syndrome
Department of Nursing, Puducherry University, Sri Balaji Vidyapeeth University, Puducherry, Tamil Nadu, India
Corresponding Author: Vijayanandh Mani, Department of Nursing, Puducherry University, Sri Balaji Vidyapeeth University, Puducherry, Tamil Nadu, India, Phone: +91 9524244889, e-mail: vanandh2010@gmail.com
How to cite this article Mani V. Lipodystrophy Syndrome. Pon J Nurs 2020;13(4):89–92.
Source of support: Nil
Conflict of interest: None
ABSTRACT
Inappropriate deposition of fat leads to different forms of diseases, causing morbidity and mortality. The person looks thin despite taking adequate lipid that leads to deposition disorder called lipodystrophy syndrome (LDS). This condition abnormally distributes the fat, where it is not supposed to be deposited. Hence, the patient looks thin and the fats are not in the subcutaneous layer of the skin. It is an autoimmune disease and caused by repeated injection of fat to a particular site. It is classified into two types: generalized and localized lipodystrophy syndrome. Treatment for this syndrome is meterleptin hormone replacement therapy and circulating the site of injection. The prognosis will vary based on the severity of the syndrome.
Keywords: Diabetes mellitus, Lipodystrophy syndrome, Morbidity, Mortality, Subcutaneous layer.
INTRODUCTION
Rare diseases are not treated frequently because healthcare professionals are not exposed to the diseases, which leads to misdiagnosis of the patient condition and treatment to the patients.1 Lipodystrophy syndrome (LDS) is a type of lipid storage disease that causes abnormal distribution of lipid in the body. This disease occurs as either genetical (congenital) or acquired (infection, auto-immune, trauma and pressure to a part of body) forms.2
DEFINITION
Lipodystrophy syndrome is a genetic or acquired disorder in which body is unable to produce and maintain healthy fat tissue.3 It is a loss of body fat from all over the body. The disorder can vary from small area of body to total absence in the whole body.4
PREVALENCE
From February 2007 to April 2009, the prevalence of LDS was 60.7%, with 22.7% having lipodystrophy, 51.7% having dyslipidemia, and 22.7% having mixed pattern in India among HIV and ART patients. The prevalence of lipodystrophy and dyslipidemia is increased among patients with HIV infection on generic ART in rural south India.5
CAUSES
The causes are:
- Measles
- Pneumonia
- Hepatitis
- Autoimmune diseases
- Repeated injection and pressure to particular body site, and
- Any injury.
TYPES
Based on how the person has LDS, it is classified as follows:
- Congenital generalized lipoatrophy
- Familial partial lipodystrophy, and
- Acquired lipoatrophy syndromes
- Localized lipodystrophy
- HAART-induced lipodystrophy in HIV-infected patients (LD-HIV)3,6
Congenital Generalized Lipoatrophy
It is an autosomal recessive disorder, where during infancy there is no fat in the subcutaneous layer and progresses to insulin resistance and diabetes mellitus during adolescence leading to pancreatitis and increased metabolic rate7 (Fig. 1).
Familial Partial Lipodystrophy
It is an autosomal dominant disorder difficult to differentiate in the affected and unaffected person before puberty. Before puberty the individual has normal fat distribution at birth and loss of subcutaneous lipid in extremities and trunk after puberty. During puberty, adequate fat will raise in neck and face8,9 (Fig. 2).
Acquired Lipoatrophy Syndrome
This occurs as the person is exposed to acute illness, which leads to increase in immune diseases that progress to autoimmune diseases. More amount of fat loss leads to drastic change in physical features10 (Fig. 3).
Localized Lipodystrophy
It occurs due to periodic injection of different drugs in subcutaneous layer at particular site. In this, the subcutaneous layer fat is lost from the focal region without affecting other layers of the skin4 (Fig. 4).
Lipodystrophy HIV
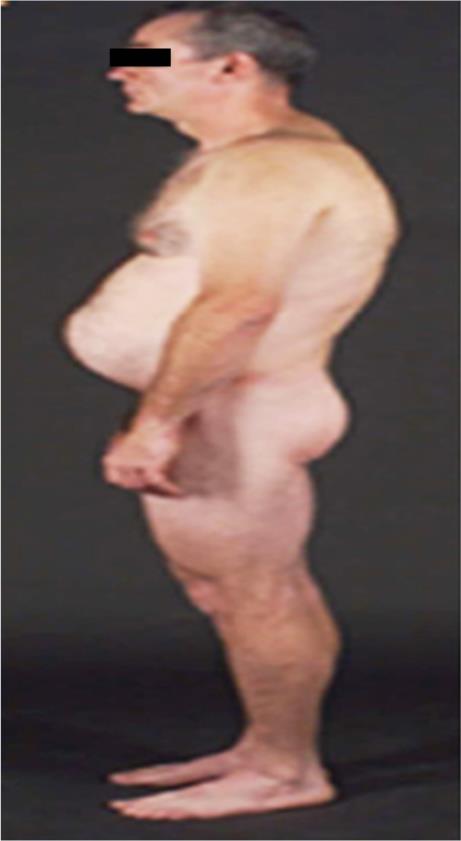
The patient who takes protease inhibitor-containing ART for >2 years leads to lose subcutaneous fat in extremities and face. Some will have buffalo hump and increased waist due to abnormally collection of fat in those areas4 (Fig. 5).

Fig. 1: An 8-year-old child with congenital LDS, child lost SC fat at birth and enlarged mandible and extremities. Child has velvety patches and dark skin patches in axillae and neck during childhood

Fig. 2: A 65-year-old female had lost subcutaneous fat in limbs during puberty and later in anterior trunk. Breast atrophy and increased fat deposited in face anterior neck vulvar and suprapubic and knees

Fig. 3: Child at 3 years (left) with normal fat distribution and at 6 years (right) lost her fat due to illness

Fig. 4: The patient has local LDS where indicated
PATHOGENESIS
The fat is deposited in the nonadipose tissue due to insufficient capacity of the subcutaneous adipose tissue. Patient will have hypertriglyceridemia, fatty liver, and liver failure, which is leading cause of death, with average lifespan of 30 years. The leptin levels are low, and patient are obese, which is associated with nonalcoholic fatty liver disease.12,13
HOW IS THE PATIENT?
The clinical presentation of patient with LDS comprises:
- Accelerated linear growth
- Increased metabolic rate
- Advanced bone age
- Belly protruded out
- Early puberty
- Mental retardation
- Insulin resistance
- Fatty liver
- Diabetes
- Cholesterol,
- Pancreatitis, and
- Heart diseases.14
HOW TO FIND THIS?
- History collection: It reveals the signs and symptoms that can be correlated with LDS.
- Physical examination: Based on the observation and assessment, the defects can be ruled out and the condition can be detected.
- Skin-fold measurement: The patient’s skin-fold thickness will be reduced compared to the other parts of the body.
- Dual-energy X-ray: Skin-fold thickness measurement with high technology
- Serum level of leptin will be less in LDS patient (normal range: 2.5–21.8 ng/mL).15
HOW TO HANDLE?
Currently, there is no treatment to cure or regrow the adipose tissue. But some of the methods to handle the condition are as follows.
Meterleptin
It is a leptin hormone replacement given as first-line treatment for metabolic and endocrine problems. The dose is based on the patient’s conditions, and every quarterly or half-yearly after laboratory investigation the dosages are altered.
- HDL cholesterol is not altered, but LDL and total cholesterol are altered in patient taking meterleptin.
- It reduces hepatic steatosis.
- Meterleptin decreased proteinuria.
Side Effects
- Hypoglycemia
- Local reaction at injection site
- T-cell lymphoma in patients
- Pancreatitis
- Liver disease
- Kidney disease.
Exercise
- Patients with LDS should be encouraged to exercise.
- Patients with cardiomyopathy should get consultation before starting exercise.
- Complicated patients with liver function should not participate in sports.
Diet
LDS patient should follow the diet:
- To control metabolic abnormalities.
- To avoid overfeeding to prevent hepatic steatosis.
- To maintain low weight in proportion to length of the body.
- Patient should take 50–60% carbohydrate, 20–30% fat, and 20% protein in the diet.
- In pancreatitis, low-protein diet is suggested.
Additional Treatments for Specific Comorbidities
Diabetes
- Metformin is a first-line agent for diabetes and insulin resistance.
- Thiazolidinediones will cause metabolic complication.
Hypertension
Angiotensin-converting enzyme inhibitors.
Contraception and Hormone Replacement Therapy
- Oral estrogens are contraindicated. If estrogen replacement is needed, transdermal estrogen should be used.
- If contraception is needed, progestin-only or nonhormonal contraceptives should be considered.16
COMPLICATIONS
The complications of the LSA are:
- Insulin resistance
- Diabetes mellitus
- Hypertriglyceridemia
- Acute pancreatitis
- Hepatic steatosis, and
- Cirrhosis.11
PROGNOSIS
Based on the severity of LDS and underlying disease such as diabetes, hypertension, etc., prognosis of the patient condition will alter, and the treatment process is different from patient to patient with the prognosis of the LDS condition.4
ROLE OF THE NURSE
LDS is rare disease, hence collaborative approach is needed with residence, nutritionist, clinical nutritionist, physiotherapist, endocrinologist, psychiatrist, and nurse and should focus on family education and individual coping strategies.
CONCLUSION
Lipodystrophy syndrome is fat deposition disorder where the patient has no fat stored in the adipose tissue and is of different type based on occurrence of the disease. There is no treatment or cure for LDS. Hormone replacement therapy with meterleptin, a substitute for leptin hormone which is less in LDS patient, dietary control, and exercise are suggested to limit the complication and lead a healthy life.
REFERENCES
1. Araújo-Vilar D, Santini F. Diagnosis and treatment of lipodystrophy: a step-by-step approach. J Endocrinol Invest 2019;42(1):61–73. DOI: 10.1007/s40618-018-0887-z.
2. “Lipodystrophy: Symptoms, Types (Familial, HIV), Causes and Treatment.” MedicineNet, Accessed 6 Apr. 2020. https://www.medicinenet.com/acquired_generalized_and_inherited_lipodystrophy/article.htm.
3. “UCLA/VA Researchers discover fat gene.”Archived from the original on 2018-10-06. Retrieved 2017-06-15.
4. “Lipodystrophy Disorders.”NORD (National Organization for Rare Disorders), https://rarediseases.org/physician-guide/lipodystrophy-disorders/ .Accessed 6 Apr. 2020.
5. Kalyanasundaram AP, Jacob SM, Hemalatha R, Sivakumar MR. Prevalence of Lipodystrophy and dyslipidemia among patients with HIV infection on generic ART in rural south india. J Int Assoc Physicians AIDS Care (Chic) 2011;11(5):329–334. DOI: 10.1177/1545109711401750.
6. Generalized Lipodystrophy: Practice Essentials, Overview, Lipodystrophy, Lipoatrophy, and Lipoatrophic Diabetes. Nov. 2019. eMedicine, https://emedicine.medscape.com/article/128355-overview.
7. Medscape, J Clin Endocrinol Metab—Content Listing. https://reference.medscape.com/viewpublication/6441 .Accessed 6 Apr. 2020.
8. Medscape, Humangenetik—Publication Information. https://reference.medscape.com/viewpublication/5968 .Accessed 6 Apr. 2020.
9. Medscape, Muscle Nerve—Publication Information. https://reference.medscape.com/viewpublication/7740 .Accessed 6 Apr. 2020.
10. Garg A. Lipodystrophies: genetic and acquired body fat disorders. J Clin Endocrinol Metab 2011;96:3313–3325. DOI: 10.1210/jc.2011-1159.
11. Herranz P, de Lucas R, Pérez-España L, Mayor M. Lipodystrophy syndromes. Dermatol Clin 2008;26(4):569–578. DOI: 10.1016/j.det.2008.05.004.
12. Polyzos SA, Perakakis N, Mantzoros CS. Fatty liver in Lipodystrophy: a review with a focus on therapeutic perspectives of adiponectin and/or leptin replacement. Metabolism 2019;96:66–82. DOI: 10.1016/j.metabol.2019.05.001.
13. Bruder-Nascimento T, Kress TC, Belin de Chantemele EJ. Recent advances in understanding Lipodystrophy: a focus on Lipodystrophy-associated cardiovascular disease and potential effects of leptin therapy on cardiovascular function. F1000Res 2019;8:F1000 Faculty Rev-1756. DOI: 10.12688/f1000research.20150.1.
14. PMC E. Europe PMC [Internet]. Europepmc.org. 2020 [cited 9 June 2020]. Available from: https://europepmc.org/article/PMC/6798323.
15. Guillin-Amarelle C, Sanchez-Iglesias S, Castro-Pais A, Rodriguez-Canete L, Ordonez-Mayan L, Pazos M, et al. Type II familial partial lipodystrophy: understanding the Kobberling syndrome. Endocrine 2016;54(2):411–421. DOI: 10.1007/s12020-016-1002-x.
16. Brown RJ, Araujo-Vilar D, Cheung PT, Dunger D, Garg A, Jack M, et al. The diagnosis and management of lipodystrophy syndromes: a multi-society practice guideline. J Clin Endocrinol Metab 2016;101(12):4500–4511. DOI: 10.1210/jc.2016-2466 academic.oup.com.
________________________
© The Author(s). 2020 Open Access This article is distributed under the terms of the Creative Commons Attribution 4.0 International License (https://creativecommons.org/licenses/by-nc/4.0/), which permits unrestricted use, distribution, and non-commercial reproduction in any medium, provided you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The Creative Commons Public Domain Dedication waiver (http://creativecommons.org/publicdomain/zero/1.0/) applies to the data made available in this article, unless otherwise stated.